Research Article | DOI: https://doi.org/10.31579/2641-5194/005
Fenofibrate Niosomes by Modified Ether Injection Method- of Proper Diet to Help Lower “Bad
Department of Gastroenterology, Bomma college pharmacy, khammam, India.
*Corresponding Author: Garrepalli samatha. Department of Gastroenterology, Bomma college pharmacy, khammam, India.
Citation: Garrepalli samatha, Uma Rani. G, Fenofibrate Niosomes by Modified Ether Injection Method- of Proper Diet to Help Lower “Bad" Cholesterol and Fats, J. Gastroenterology Pancreatology and Hepatobilary Disorders. 2(1). DOI: 10.31579/2641-5194/005
Copyright: © 2018 Garrepalli samatha. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 22 September 2018 | Accepted: 19 October 2018 | Published: 07 November 2018
Keywords: fenofibrate niosomes; ether injection method; proper diet
Abstract
The quest never ends. From the very beginning of the human race the quest is going on for newer and better alternatives and in case of drugs it will continue till we find a drug with maximum efficacy and no side effects. Many drugs, particularly chemotherapeutic agents, have narrow therapeutic window and their clinical uses are limited and compromised by dose limiting toxic effect. Thus, the therapeutic effectiveness of the existing drugs is improved by formulating them in an advantageous way.
In the past few decades, considerable attention has been focused on the development of new drug delivery system (NDDS). The NDDS should ideally full fill two prerequisites. Firstly, it should deliver the drug at a rate directed by the needs of the body, over the period of treatment. Secondly, it should channel the active entity to the site of action. Conventional dosage forms including prolonged release dosage forms are unable to meet none of these. At present, no available drug delivery system behaves ideally, but sincere attempts have been made to achieve them through various novel approaches in drug delivery1. Approaches are being adapted to achieve this goal, by paying considerable attention either to control the distribution of drug by incorporating it in a carrier system, or by altering the structure of the drug at the molecular level, or to control the input of the drug into the bio environment to ensure an appropriate profile of distribution.
Introduction
Novel drug delivery system aims at providing some control, whether this is of temporal or spatial nature, or both, of drug release in the body. Novel drug delivery attempts to either sustain drug action at a predetermined rate or by maintaining a relatively constant, effective drug level in the body with concomitant minimization of undesirable side effects. It can also localize drug action by spatial placement of controlled release systems adjacent to, or in the diseased tissue or organ or target drug action by using carriers or chemical derivatization to deliver drug to particular target cell type.
Vesicular systems-A Review
In recent years, vesicles have become the vehicle of choice in drug delivery. Lipid vesicles were found to be of value in immunology, membrane biology, diagnostic techniques and most recently, genetic engineering7-9. Vesicles can play a major role in modeling biological membranes and in the transport and targeting of active agents. Encapsulation of a drug in vesicular structures can be predicted to prolong the existence of the drug in systemic circulation and perhaps, reduces the toxicity if selective uptake can be achieved10. The phagocytic uptake of the systemic delivery of drug loaded vesicular delivery system provides an efficient method for delivery of drug directly to the site of infection, leading to reduction of drug toxicity with no adverse effects. Vesicular drug delivery reduces the cost of therapy by improved bioavailability of medication, especially in case of poorly soluble drugs. They can incorporate both hydrophilic and lipophilic drugs. These systems delay drug elimination of rapidly metabolizable drugs and function as sustained release systems and solve the problems of drug insolubility, instabilityand rapid degradation. Consequently, a number of vesicular delivery systems such as liposomes,transfersomes, pharmacosomes, niosomesetc, were developed.
Liposomes
Liposomes are simple microscopic vesicles in which lipid bilayer structures are present with an aqueous volume entirely enclosed by a membrane, composed of lipid molecule. There are a number of components present in liposomes, with phospholipid and cholesterol being the main ingredients. The type of phospholipids includes phosphoglycerides and sphingolipids and together with their hydrolysis products.
Niosomes
Niosomes or nonionic surfactant vesicles are microscopic lamellar structures formed on admixture of nonionic surfactant of the alkyl or di alkyl polyglycerolether class and cholesterol with subsequent hydration in aqueous media. In niosomes, the vesicles forming amphiphile is a nonionic surfactant such as span 60 which is usually stabilized by addition of cholesterol and small amount of anionic surfactant such as dicetyl phosphate.Niosomes can entrap both hydrophilic and lipophilic drugs, either in aqueous layer or in vesicular membrane made of lipid materials. It is reported to attain better stability than liposomes. It can prolong the circulation of the entrapped drugs. Because of the presence of nonionic surfactant with the lipid, there is better targeting of drugs to tumour, liver and brain.
Methodology
Analytical methods reported for fenofibrate
Fenofibrate is a non steroidal antiinflammatory drug with good analgesic and antirheumatic properties. Chemically it is [[[2-[(2, 6-Dichlorophenyl) amino] phenyl] acetyl] oxy] acetic acid. It is used in various pain conditions like rheumatoid arthritis, osteoarthritis and ankylosing spondylatis. It is of ficial in British Pharmacopoeia. Several analytical techniques like titrimetric, colourimetric,spectroflurimetric ,densitometric,HPLC, RP-HPLC, spectrophotometric and stripping voltametric have been reported for the estimation of fenofibrate.
Zawilla et al studied three sensitive and reproducible methods for quantitative determination of fenofibrate in pure form and in pharmaceutical formulation are presented. The first method is based on the reaction between the drug via its secondary aromatic amino group and p-dimethyl amino cinnamaldehyde (PDAC) in acidified methanol to give a stable coloured complex after heating at 75°C for 20 min. Absorption measurements were carried out at 665.5nm. Beer’s law is obeyed over concentration range 20-100µ g/ml with mean recovery 100.33±0.84. The other two methods are high performance liquid chromatography (HPLC) and densitometric methods by which the drug was determined in the presence of its degradation products over concentration range of 20-70ng/ml and 1-10ng/spot and mean recoveries are 99.59±0.90 and 99.45±1.09 respectively.
Rohit Shah et al developed a new, precise and simple UV spectrophotometric method for the estimation of fenofibrate from tablet formulation. The drug obeyed the Beer’s law and showed good correlation.
Preparation of niosomes
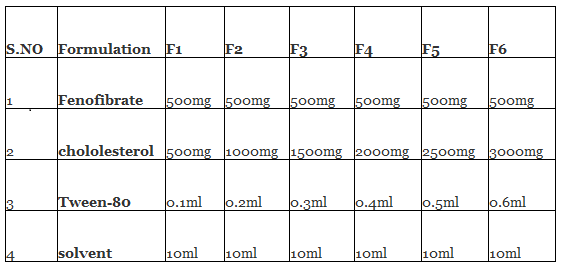
Modified Ether Injection process: Niosomes containing fenofibrate were prepared by modified ether injection technique using nonionic surfactants (spans and tweens) and cholesterol at different concentrations. Cholesterol and surfactant were dissolved in 6ml diethyl ether mix Ether Injection values with 2ml methanol which previously containing weighed quantity of fenofibrate. Then, the resulting solution was slowly injected using microsyringe at a rate of 1ml/min into 15 ml of hydrating solution (phosphate buffer pH 7.4).The solution was stirred continuously on magnetic stirrer and temperature is maintained at 60-65ºC. As the lipid solution was injected slowly into aqueous phase, the differences in temperature between phases cause rapid vaporization of ether results in spontaneous vesiculation and formation of niosomes.
FORMULATION OF FENOFIBRATE NIOSOMES:
Results
Determination of absorption maxima: Absorption maxima or the wavelength at which absorption takes place. For accurate analytical work it is important to determine the absorption maxima of the substance under study.
Method: UV method
Equipments: UV-VIS spectrophotometer
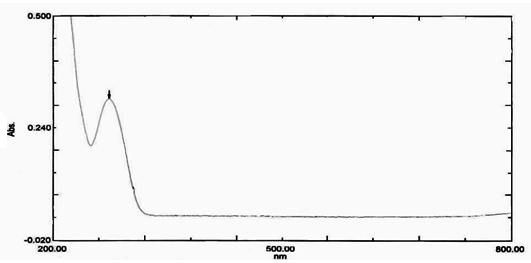
100mg of fenofibrate was dissolved in 100ml dried methanol. 1ml of this solution was pipetted out in separate volumetric flask and diluted with phosphate buffer 7.4 and subjected for UV scanning in the range of 200-800 using Double beam UV-VIS spectrophotometer, (pharmaspec-1700,shimadzu, japan). The absorption maxima obtained at 274 with a characteristic peak (figure 1)
Conclusion
Niosomes containing fenofibrate were conveniently prepared by modified ether injection technique using nonionic surfactants (tween 80) and cholesterol at different concentrations.
All the niosome formulations were subjected for entrapment efficiency and the results shows that as the concentration of surfactant increases the entrapment efficiency increases tween 80 The niosomal formulations of span 60 with various concentrations of cholesterol shows that lower the cholesterol concentration higher the entrapment efficiency but in case of tween 80 as the concentration of cholesterol increases the entrapment efficiency increases.
References
- Bornstein, R. F. (2011). Toward a multidimensional model of personality disorder diagnosis: Implications for DSM–5. Journal of Personality Assessment, 93(4), 362-369.
View at Publisher | View at Google Scholar - Cramer, P. (1998a). Defensiveness and defense mechanisms. Journal of Personality, 66, 879–894. doi:10.1111/1467-6494.00035
View at Publisher | View at Google Scholar - Eysenck, H. J. (1994). Normality-abnormality and the three-factor model of personality. Differentiating normal and abnormal personality (pp. 3–25). New York: Springer.
View at Publisher | View at Google Scholar - Goldberg, L. R. (1993). The structure of phenotypic personality traits. American Psychologist, 48, 26–34.
View at Publisher | View at Google Scholar - Jang, K. L., & Livesley, W. J. (1999). Why do measures of normal and disordered personality correlate? A study of genetic comorbidity. Journal of Personality Disorders, 13, 10–17.
View at Publisher | View at Google Scholar - Kramer, U., Roten, Y, Perry, J. C. & Despland J. N. (2013). Beyond splitting: Observer-rated d efense mechanisms in borderline personality disorder. Psychoanalytic Psychology. Vol. 30, No. 1, 3–15
View at Publisher | View at Google Scholar - Krueger, R. F. & Markon, K. E. (2005). Delineating the Structure of Normal and Abnormal Personality: An Integrative Hierarchical Approach University of Minnesota. Journal of Personality and Social Psychology. Vol. 88, No. 1, 139–157.
View at Publisher | View at Google Scholar - Livesley, W. J. (2001). Commentary on reconceptualizing personality disorder categories using trait dimensions. Journal of Personality, 69, 277–286.
View at Publisher | View at Google Scholar - Markon, K. E., Krueger, R. F., Bouchard, T. J., & Gottesman, I. I. (2002). Normal and abnormal personality traits: Evidence for genetic and environmental relationships in the Minnesota Study of Twins Reared Apart. Journal of Personality, 70, 661–693.
View at Publisher | View at Google Scholar - O’Connor, B. P., & Dyce, J. A. (2001). Rigid and extreme: A geometric representation of personality disorders in five-factor model space. Journal of Personality and Social Psychology, 81, 1119–1130.
View at Publisher | View at Google Scholar - O’Connor, B. P. (2002). The search for dimensional structure differences between normality and abnormality: A statistical review of published data on personality and psychopathology. Journal of Personality and Social Psychology, 83, 962–982.
View at Publisher | View at Google Scholar - Perry, J. C., Presniak, M. D., & Olson, T. R. (2013) Defense Mechanisms in Schizotypal, Borderline, Antisocial, and Narcissistic Personality Disorders. Spring. Psychiatry. 76 (1). 45-48.
View at Publisher | View at Google Scholar - Presniak, M. D., Olson, T. R., & Macgregor M. W. (2010). The Role of Defense Mechanisms in Borderline and Antisocial Personalities. Journal of Personality Assessment, 92 (2), 137–145.
View at Publisher | View at Google Scholar - Samuel, D. B., & Widiger, T. A. (2004). Clinicians' descriptions of prototypic personality disorders. Journal of Personality Disorders, 18, 286-308.
View at Publisher | View at Google Scholar - Samuel, D. B., & Widiger, T. A. (2008). A Meta-Analytic Review of the Relationships Between the Five- Factor Model and DSM-IV-TR Personality Disorders: A Facet Level Analysis University of Kentucky. Clinical Psychology Review. December (8), 1326–1342. doi:10.1016/j.cpr.2008.07.002.
View at Publisher | View at Google Scholar - Samuel, D. B., Lynam, D. R., Widiger, T., & Ball, S. A. (2012). An expert consensus approach to relating the proposed DSM-5 types and traits. Department of Psychological Sciences Faculty Publications. Paper 13. http://dx.doi.org/10.1037/a0023787
View at Publisher | View at Google Scholar - Vahia, V. N. (2013). Diagnostic and statistical manual of mental disorders 5: A quick glance. Indian journal of psychiatry, 55(3), 220.
View at Publisher | View at Google Scholar - Vaillant, G. E. (1992). Ego mechanisms of defense: A guide for clinicians and researchers. Washington, DC., American Psychiatric Press.
View at Publisher | View at Google Scholar - Widiger, T. A., & Costa, P. T. (1994). Personality and personality disorders. Journal of Abnormal Psychology, 103, 78–91.
View at Publisher | View at Google Scholar - Widiger, T. A., & Trull, T. J. (2007). Plate tectonics in the classification of personality disorder: shifting to a dimensional model. American Psychologist, 62, 71-83.
View at Publisher | View at Google Scholar
Dear Grace Pierce, Editorial Coordinator of Journal of Clinical Research and Reports, Thank you for the speedy and efficient peer review process. I appreciate the fact that your peer reviewers do not take months to respond like with some other journals. I would also like to thank the editorial office for responding quickly to my questions. It is an excellent journal. I plan to submit more manuscripts in the future. Best wishes from, Robert W. McGee

Robert W McGee
Dear Grace Pierce, Editorial Coordinator of Journal of Clinical Research and Reports, Working with you and your team on our recent publication in JCRR has been a truly wonderful and enjoyable experience. The responses were prompt, and the reviewers were patient, constructive, and highly professional. One reviewer in particular gave me the feeling that a professor was carefully reading and commenting on my coursework, which was deeply touching. The entire process was straightforward and hassle‑free, with no tedious online forms to complete. I highly recommend this journal. Best wishes from, DR Aibing Rao, Head of R&D

Aibing Rao
I Appreciate the Opportunity to Share my Experience with the Journal of Clinical Research and Reports. The peer review process was timely and constructive, and the feedback provided helped improve the quality of our manuscript. The editorial office was professional, responsive, and supportive throughout the process, ensuring smooth communication and efficient handling of the submission. Overall, it was a positive experience collaborating with your team.

Kashani Mehdi
Dear Mercy Grace, Editorial Coordinator of Obstetrics Gynecology and Reproductive Sciences, We would like to express our gratitude for your help at all stages of publishing and editing the article. The editors of the magazine answer all the necessary questions and help at every stage. We will definitely continue to cooperate and publish other works in the Obstetrics Gynecology and Reproductive Sciences! Best wishes from, Alla Konstantinovna Politova,

Alla Konstantinovna Politova
Dear Maria Emerson, Editorial Coordinator of International Journal of Clinical Case Reports and Reviews, What distinguishes International Journal of Clinical Case Report and Review is not only the scientific rigor of its publications, but the intellectual climate in which research is evaluated. The submission process is refreshingly free of unnecessary formal barriers and bureaucratic rituals that often complicate academic publishing without adding real value. The peer-review system is demanding yet constructive, guided by genuine scientific dialogue rather than hierarchical or authoritarian attitudes. Reviewers act as collaborators in improving the manuscript, not as gatekeepers imposing arbitrary standards. This journal offers a rare balance: high methodological standards combined with a respectful, transparent, and supportive editorial approach. In an era where publishing can feel more burdensome than research itself, this platform restores the original purpose of peer review — to refine ideas, not to obstruct them Prof. Perlat Kapisyzi, FCCP PULMONOLOGIST AND THORACIC IMAGING.
Perlat Kapisyzi
Dear Reader: We have published several articles in the Auctores Publishing, LLC, journal, Clinical Medical Reviews and Reports in recent years (CMRR). This is an ‘open access’ journal and the following are our observations. From the initial invitation to submit an article, to the final edits of galley proofs, we have found CMRR personnel to be professional, responsive, rapid and thorough. This entire process begins with Catherine Mitchell, Editorial Coordinator. She is simply outstanding, and, I believe, unparalleled in her capacity. I cannot imagine a more responsive and dedicated Editorial Coordinator. As I read the dates and timing of her correspondence with us, it seems that she never sleeps. I hope Auctores Publishing, LLC, appreciates her efforts as much as these authors do. Thank you to Auctores Publishing, LLC, to the Editorial Staff/Board, and to Catherine Mitchell from a grateful author(s).