Research Article | DOI: https://doi.org/10.31579/2693-7247/055
Design and Optimizations of Aceclofenac Bioadhesive Extended Release Microspheres
1 M. Pharm., Ph. D, Associate Professor, Department of Pharmaceutics, Unity College of Pharmacy, Telangana, India.
2 B. Pharm, Unity College of Pharmacy, Telangana, India.
*Corresponding Author: G. Prasad M. Pharm., Ph. D, Associate Professor, Department of Pharmaceutics, Unity College of Pharmacy, Telangana, India.
Citation: G. Prasad, K. Devika, P. Varshith, B. Shravani, E. Pavithra and Ch. Swathi (2021) Design and Optimizations of Aceclofenac Bioadhesive Extended Release Microspheres Development; J. Pharmaceutics and Pharmacology Research 4(4) DOI: 10.31579/2693-7247/055
Copyright: © 2021, G. Prasad, This is an open access article distributed under the Creative Commons Attribution License, which permit unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 27 October 2021 | Accepted: 02 November 2021 | Published: 13 November 2021
Keywords: microspheres; zaltoprofen; aceclofenac
Abstract
The oral route for drug delivery is the most popular, desirable, and most preferred method for administrating therapeutically agents for systemic effects because it is a natural, convenient, and cost effective to manufacturing process. Oral route is the most commonly used route for drug administration. Although different route of administration are used for the delivery of drugs, oral route remain the preferred mode. Even for sustained release systems the oral route of administration has been investigated the most because of flexibility in designing dosage forms.
Present controlled release drug delivery systems are for a maximum of 12 hours clinical effectiveness. Such systems are primarily used for the drugs with short elimination half life.
1. Introduction
For many decades, medication of an acute disease or a chronic disease has been accomplished by delivering drugs to the patients via various pharmaceutical dosage forms like tablets, capsules, pills, creams, ointments, liquids, aerosols, injectable and suppositories as carriers [1]. To achieve and then to maintain the concentration of drug administered within the therapeutically effective range needed for medication, it is often necessary to take this type of drug delivery systems several times in a day. This results in a fluctuated drug level and consequently undesirable toxicity and poor efficiency. This factor as well as other factors such as repetitive dosing and unpredictable absorption leads to the concept of controlled drug delivery systems [2, 3]. The word new or novel in the relation to drug delivery system is a search for something out of necessity. An appropriately designed sustained or controlled release drug delivery system can be major advance toward solving the problem associated with the existing drug delivery system.
The objective of controlled release drug delivery includes two important aspects namely spatial placement and temporal delivery of drug. Spatial placement relates to targeting a drug to a specific organ or tissue, while temporal delivery refers to controlling the rate of drug delivery to the target tissue.
Oral controlled release dosage forms have been developed over the past three decades due to their considerable therapeutic advantages such as ease of administration, patient compliance and flexibility in formulation. However, this approach is be filled with several physiological difficulties such as inability to restrain and locate the controlled drug delivery system within the desired region of the gastrointestinal tract (GIT) due to variable motility and relatively brief gastric emptying time (GET) in humans which normally averages 2-3 h through the major absorption zone, i.e., stomach and upper part of the intestine can result in incomplete drug release from the drug delivery system leading to reduced efficacy of the administered dose.
The objective in designing a controlled release system is to deliver the drug at a rate necessary to achieve and maintain a constant drug blood level. This rate should be similar to that achieved by continuous intravenous infusion where a drug is provided to the patient at a rate just equal to its rate of elimination. This implies that the rate of delivery must be independent of the amount of drug remaining in the dosage form and constant over time, i.e., release from the dosage form should follow zero-order kinetics.
2. Aim and Objective
- Aim of the study is to formulate Aceclofenac bioadhesive extended release microspheres using different polymers.
- To formulate the Aceclofenac bioadhesive extended release microspheres using different polymers like sodium alginate, Guar Gum, Locust Bean Gum, Xanthan Gum.
- To choose the better formulation among the prepared formulations which shows better release and bioadhesion.
Preformulation Studies
Spectroscopic Studies
Preparation of 0.1N HCl (pH 1.2)
Take 8ml of HCl in a 1000ml volumetric flask and make up the volume with distilled water
Determination of λMax:
Stock solution (1000µg/ml) of Aceclofenac was prepared in methanol. This solution was appropriately diluted with 0.1N HCl(pH 1.2) and 6.8 pH phosphate buffer to obtain a concentration of 10µg/ ml. The resultant solution was scanned in the range of 200nm to 400nm on UV-Visible spectrophotometer. The drug exhibited a λmax at 252nm and 254nm.
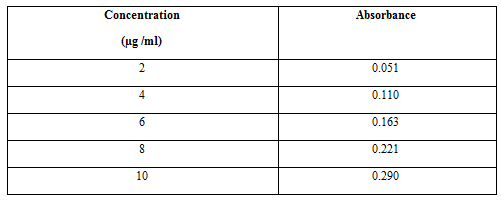
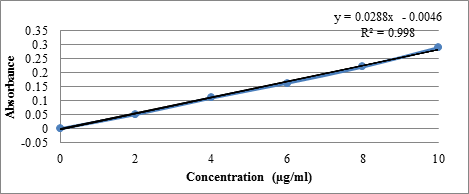
Preparation of Standard Calibration Curve of Aceclofenac:
- 10 mg of Aceclofenac was accurately weighed and dissolved in 10ml of methanol (Stock Solution–I) to get a concentration of 1000 μg/ml.
- From the stock solution- I,1ml of aliquots was taken and suitably diluted with 0.1N HCl (Stock Solution-II) to get concentrations of 100μg/ml.
- From the stock solution- II,aliquots were taken and suitably diluted with 0.1N HCl (pH 1.2) to get concentrations in the range of 2 to 10μg/ml.The absorbance of these samples were analyzed byusing UV-Visible Spectrophotometer at 252nm against reference solution 0.1N HCl (pH 1.2).The same procedure is repeated with 6.8pH phosphate buffer also
The Linear Regression Analysis:
The linear regression analysis was done on Absorance points. The standardcalibration curve obtained had a Correlation Coefficient of 0.998 with of slope of 0.028 and intercept of 0.004.
Compatibility Studies
A proper design and formulation of a dosage form requires considerations of the physical, chemical and biological characteristics of both drug and excipients used in fabrication of the product. Compatibility must be established between the active ingredient and other excipients to produce a stable, efficacious, attractive and safe product. If the excipient(s) are new and if no previous literature regarding the use of that particular excipient with an active ingredient is available, then compatibility studies are of paramount importance. Hence, before producing the actual formulation, compatibility of Aceclofenac with different polymers and other excipients was tested using the Fourier Transform Infrared Spectroscopy (FT-IR) technique.
Fourier Transform Infrared Spectroscopy (Ft-Ir):
In order to check the integrity (Compatibility) of drug in the formulation,FT-IR spectra of the formulations along with the drug and other excipients were obtained and compared using Shimadzu FT-IR 8400 spectrophotometer. In the present study, Potassium bromide(KBr) pellet method was employed. The samples were thoroughly blended with dry powdered potassium bromide crystals. The mixture was compressed to form a disc. The disc was placed in the spectrophotometer and the spectrum was recorded.The FT-IR spectra of the formulations were compared with the FT-IR spectra of the pure drug and the polymers.
3. Method of Preparation
Ionotropic Gelation Method:
Batches of microcapsules were prepared by ionotropic gelation method which involved reaction between sodium alginate and polycationic ions like calcium to produce a hydrogel network of calcium alginate. Sodium alginate and the mucoadhesive polymer were dispersed in purified water (10 ml) to form a homogeneous polymer mixture. The API, Aceclofenac (100 mg) were added to the polymer premix and mixed thoroughly with a stirrer to form a viscous dispersion. The resulting dispersion was then added through a 22G needle into calcium chloride (4% w/v) solution. The addition was done with continuous stirring at 200rpm. The added droplets were retained in the calcium chloride solution for 30 minutes to complete the curing reaction and to produce rigid spherical microcapsules. The microcapsules were collected by decantation, and the product thus separated was washed repeatedly with purified water to remove excess calcium impurity deposited on the surface of microcapsules and then air-dried.
Characterization of Microcapsules:
Percentageyield
The percentage of production yield was calculated from the weight of dried microsphe-res recovered from each batch and the sum of the initial weight of starting materials. The percentage yield was calculated using the following formula:
Practical mass (Microcapsules)
% Yield=-----------------------------------------------x100
Theoretical mass (Polymer + Drug)
Drug entrapment efficiency:
Microcapsules equivalent to 100 mg of the drug Aceclofenac were taken for evaluation. The amount of drug entrapped was estimated by crushing the microcapsules. The powder was transferred to a 100 ml volumetric flask and dissolved in 10ml of methanol and the volume was made up using simulated gastric fluid pH 1.2. After 24 hours the solution was filtered through Whatmann filter paper and the absorbance was measured after suitable dilution spectrophotometrically at 252 nm. The amount of drug entrapped in the microcapsules was calculated by the following formula,
Experimental Drug Content
% Drug Entrapment Efficiency = - - - - - - - - - - - - - - - - - - - - - - - × 100
Theoretical Drug Content
Particle size analysis:
Samples of the microparticles were analyzed for particle size by optical microscope. The instrument was calibrated and found that 1unit of eyepiece micrometer was equal to 12.5μm. Nearly about 100 Microparticles sizes were calculated under 45x magnification. The average particle size was determined by using the Edmondson’s equation:
nd
Dmean=------
n
Where,
n – Number of microcapsules observed
D – Mean size range
Swelling study:
Swelling ratio of different dried microcapsules were determined gravimetrically in simulated gastric fluid pH 1.2 .The microcapsules were removed periodically from the solution, blotted to remove excess surface liquid and weighed on balance. Swelling ratio (% w/v) was determined from the following relationship:
(Wt – W0)
Swelling ratio = - - - - - - - - - - - × 100
(W0)
Where W0 & Wt are initial weight and Final weight of microcapsules respectively.
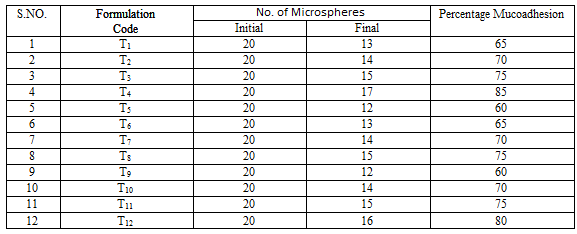
Evaluation of mucoadhesive property:
The mucoadhesive property of microcapsules was evaluated by an in vitro adhesion testing method known as wash-off method. Freshly excised pieces of goat stomach mucous were mounted on to glass slides with cotton thread. About 20 microcapsules were spread on to each prepared glass slide and immediately thereafter the slides were hung to USP II tablet disintegration test, when the test apparatus was operated, the sample is subjected to slow up and down movement in simulated gastric fluid pH 1.2 at 370C contained in a 1-litre vessel of the apparatus. At an interval of 1 hour up to 8 hours the machine is stopped and number of microcapsules still adhering to mucosal surface was counted.
Number of microcapsules adhered
% Mucoadhesion= ------------------------------------------ ×100
Number of microcapsules applied
In vitro drug release study:
The dissolution studies were performed in a fully calibrated eight station dissolution test apparatus (37 ± 0.50C, 50 rpm) using the USP type – I rotating basket method in simulated gastric fluid pH 1.2 (900ml). A quantity of accurately weighed microcapsules equivalent to 100mg Aceclofenac each formulation was employed in all dissolution studies. Aliquots of sample were withdrawn at predetermined intervals of time and analyzed for drug release by measuring the absorbance at 252nm. At the same time the volume withdrawn at each time intervals were replenished immediately with the same volume of fresh pre-warmed simulated gastric fluid pH 1.2 maintaining sink conditions throughout the experiment.
4. Results and Discussion
4.1. Preformulation Studies
4.1.1. Spectroscopic Studies
Determination of λmax
A solution of 10µg/ml of Aceclofenac was scanned in the range of 200 to 400nm. The drug exhibited a λmax at 252nm in simulated gastric fluid pH 1.2 and had good reproducibility. Correlation between the concentration and absorbance was found to be near to 0.998, with a slope of 0.028 and intercept of 0.004.
Calibration curve of Aceclofenac in simulated gastric fluid pH 1.2
Compatibility Studies
Drug polymer compatibility studies were carried out using Fourier Transform Infra-Red spectroscopy to establish any possible interaction of Drug with the polymers used in the formulation. The FT-IR spectra of the formulation were compared with the FTIR spectra of the pure drug.
Evaluation and Characterisation of Microspheres
Percentage Yield
It was observed that as the polymer ratio in the formulation increases, the product yield also increases. The low percentage yield in some formulations may be due to blocking of needle and wastage of the drug- polymer solution, adhesion of polymer solution to the magnetic bead and microspheres lost during the washing process. The percentage yield was found to be in the range of 80 to 88% for microspheres containing sodium alginate along with GUAR GUMas copolymer, 62.22 to 87% for microspheres containing sodium alginate along with LOCUST BEAN GUM as copolymer and 80 to 87.5% for microspheres containing sodium alginate along with XANTHAN GUMas copolymer. The percentage yield of the prepared microspheres is recorded in Table 6.2 and displayed in Figures 6.4 to 6.6.
Drug Entrapment Efficiency
Percentage Drug entrapment efficiency of Aceclofenac ranged from 82.66 to 88.66% for microspheres containing sodium alginate along with GUAR GUMas copolymer, 53.2 to 76.66% for microspheres containing sodium alginate along with Locust Bean Gum as copolymer and 66.73 to 79.2% for microspheres containing sodium alginate along with Xanthan Gum as copolymer. The drug entrapment efficiency of the prepared microspheres increased progressively with an increase in proportion of the respective polymers. Increase in the polymer concentration increases the viscosity of the dispersed phase. The particle size increases expone- ntially with viscosity. The higher viscosity of the polymer solution at the highest polymer concentration would be expected to decrease the diffusion of the drug into the external phase which would result in higher entrapment efficiency. The % drug entrapment efficiency of the prepared microspheres is displayed in Table 6.3, and displayed in Figures.
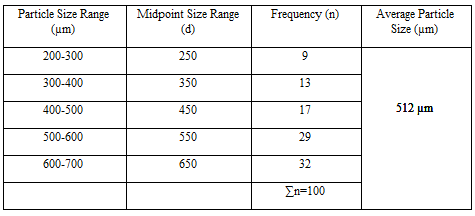
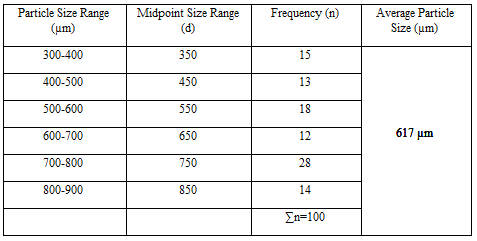
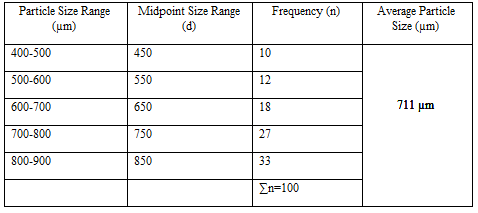
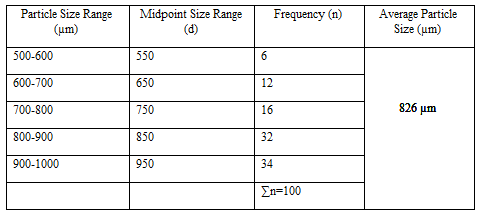
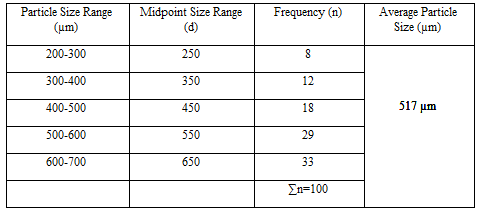
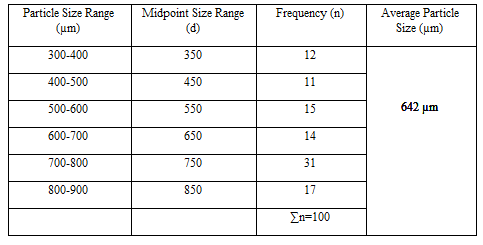
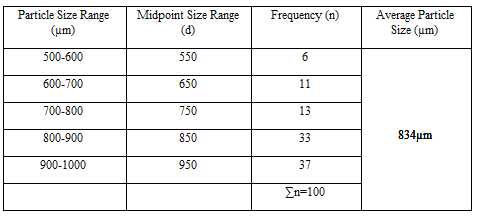
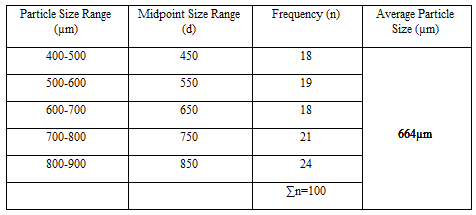
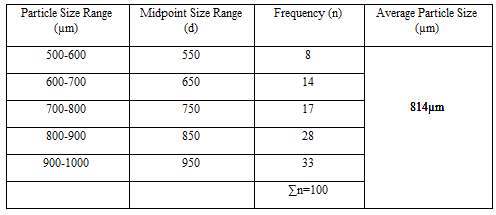
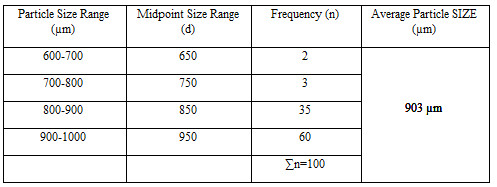
Particle Size Analysis
The mean size increased with increasing polymer concentration which is due to a significant increase in the viscosity, thus leading to an increased droplet size and finally a higher microspheres size. Microspheres containing sodium alginate along with Guar gum as copolymer had a size range of 512µm to 826µm, microspheres containing sodium alginate along with Locust Bean Gum as copolymer exhibited a size range between 517µm to 834µm and microspheres containing sodium alginate along with XANTHAN GUM as copolymer had a size range of 664µm to 903µm. The particle size data is presented in Tables 6.3 to 6.13 and displayed in Figures. The effect of drug to polymer ratio on particle size is displayed in Figure. The particle size as well as % drug entrapment efficiency of the microspheres increased with increase in the polymer concentration.
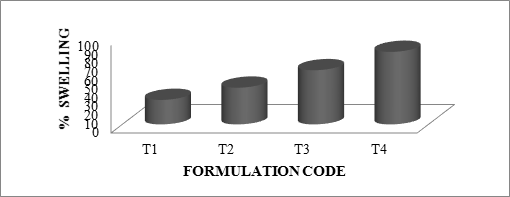
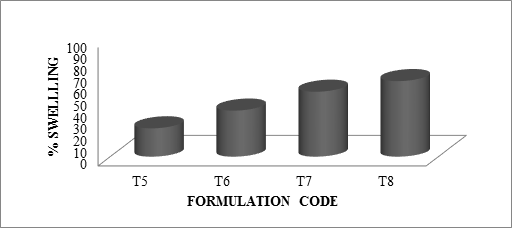
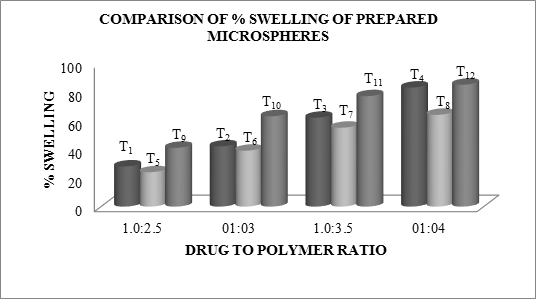
The swelling ratio is expressed as the percentage of water in the hydrogel at any instant during swelling. Swell ability is an important characteristic as it affects mucoadhesion as well as drug release profiles of polymeric drug delivery systems. Swell ability is an indicative parameter for rapid availability of drug solution for diffusion with greater flux. Swell ability data revealed that amount of polymer plays an important role in solvent transfer. It can be concluded from the data shown in Table 6.14 that with an increase in polymer concentration, the percentage of swelling also increases. Thus we can say that amount of polymer directly affects the swelling ratio. As the polymer to drug ratio increased, the percentage of swelling increased from 28 to 85% for microspheres containing sodium alginate along with GUAR Gum as copolymer, 24 to 64% for microspheres containing sodium alginate along with Locust Bean Gum as copolymer and 31 to 85 for microspheres containing sodium alginate along with Xanthan Gum as copolymer. The percentage of swelling of the prepared microspheres is displayed in Figures. The effect of drug to polymer ratio on percentage swelling is displayed in Figure.
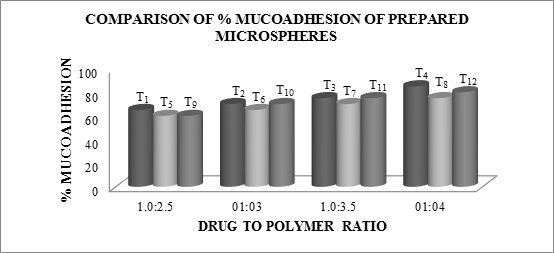
In-Vitro Mucoadhesion Test
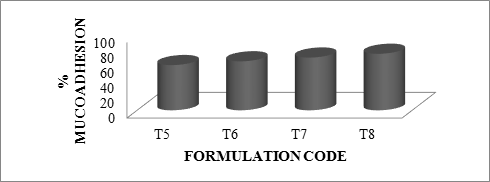
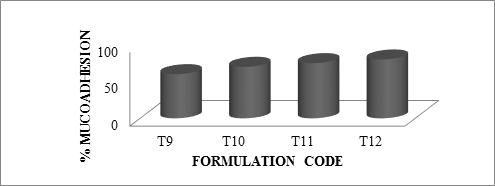
As the polymer to drug ratio increased, microspheres containing sodium alginate along with Guar Gum as copolymer exhibited % mucoadhesion ranging from 65 to 85%, microspheres containing sodium alginate along with Locust Bean Gum as copolymer exhibited % mucoadhesion ranging from 60 to 75% and microspheres containing sodium alginate along with Xanthan Gum as copolymer exhibited % mucoadhesion ranging from 60 to 80%.
The rank of order of mucoadhesion is Guar Gum> Xanthan Gum > Locust Bean Gum. The results of in-vitro mucoadhesion test are compiled in Table 6.15. Effect of polymer proportion on % mucoadhesion is depicted in Figures 6.14 to 6.16 and comparative depiction of % mucoadhesion is depicted in Figure 6.17.
In-Vitro Drug Release Studies
Dissolution studies of all the formulations were carried out using dissolution apparatus USP type I. The dissolution studies were conducted by using dissolution media, pH 1.2.for fisrt 2hr and then it was eplaced with 6.8ph phosphate buffer The results of the in-vitro dissolution studies of formulations T1 to T4, T5 to T8 and T9 to T12 are shown in table. The plots of Cumulative percentage drug release Vs Time. Figure shows the comparison of
5. Conclusion
In thepresent work, bioadhesive controlled release microspheresof Zaltoprofen using Sodium alginate along with HPMC K100M, HPMC K15M, XANTHAN GUMas copolymers wereformulated to deliver Zaltoprofen via oral route.
Details regarding the preparation and evaluation of the formulations have been discussed in the previous chapter. From the study following conclusions could be drawn:-
- The results of this investigation indicate that ionic cross linking technique Ionotropic gelation method can be successfully employed to fabricate Zaltoprofen microspheres. The technique provides characteristic advantage overconventional microsphere method, which involves an “all-aqueous” system, avoids residualsolvents in microspheres. Other methods utilize larger volume of organic solvents, which are costly and hazardous because of the possible explosion, air pollution, toxicity and difficult to remove traces of organic solvent completely.
- FT-IR spectra of the physical mixture revealed that thedrug is compatible with the polymers and copolymers used.
- Micromeritic studies revealed that the mean particle size of the prepared microspheres was in the size range of 512-903µm and are suitablefor bioadhesive controlled release microspheresfor oral administration.
- Increase in the polymerconcentration led to increase in % Yield, %Drug entrapment efficiency, Particle size, % swelling and % Mucoadhesion.
- The in-vitro mucoadhesivestudy demonstrated that microspheres of Zaltoprofen using sodium alginate along with HPMC K100M as copolymer adhered tothe mucus to a greater extent thanthe microspheres of Zaltoprofen using sodium alginate along with HPMC K15M and XANTHAN GUM as copolymers.
References
- Mulugeta Fentie, (2015). Anteneh Belete and Tsige Gebre-Mariam Formulation of Sustained Release Floating Microspheres of Furosemide from Ethylcellulose and Hydroxypropyl Methylcellulose Polymer Blends J Nanomed Nanotechnol 6:1
View at Publisher | View at Google Scholar - Abhijeet A. Durgavale, Archana R. Dhole, Shrinivas K. Mohite, Chandrakant S, (2012) . Magdum Formulation and Evaluation of Floating Microsphere of Captopril using Different Gas Forming Agents. Am. J. PharmTech Res. 2(2)
View at Publisher | View at Google Scholar - A Salomy Monica Diyya, V Rakesh Kumar,( 2015). Formulation and In Vitro Evaluation of Floating Microspheres of Glipizide, Volume 3 (2), Page-606-615 606 IIIIIIIII© International Journal of Pharma Research and Health Sciences. All rights reserved CODEN (US
View at Publisher | View at Google Scholar - Megha Sharma, Seema Kohli,, Agnimitra Dinda In-vitro and in-vivo evaluation of repaglinide loaded floating microspheres prepared from different viscosity grades of HPMC polymer.
View at Publisher | View at Google Scholar - Saudi Pharmaceutical Journal Available online 27 February 2015
View at Publisher | View at Google Scholar - Pavan Kumar Perumandla, Sree Priya. Formulation And In Vitro Evaluation Of Floating Microspheres Of Dextromethorphan Hydrobromide , International Journal of Pharmacy and Pharmaceutical Sciences ISSN- 0975-1491 Vol 6, Issue 4, 2014.
View at Publisher | View at Google Scholar - S.K.Senthilkumar, B.Jaykar, S.Kavimani, (2010). Formulation, Characterization and Invitro Evaluation of Floating Microsphere Containing Rabeprazole Sodium, /JITPS Vol.1 (6), 274-282.
View at Publisher | View at Google Scholar - V. Sai Kishore, B. Tejeswara Rao, K. Pavani, D. Nagasen, K. Rakesh Varma and D. Gowtham Plantago Ovata Seeds And Bhringaraj, (2013). Leaves As Superdisintegrants: Formulation And Evaluation Of Sotalol Hydrochloride Orodispersible Tablets IJPCBS 3(4), 1040-1046
View at Publisher | View at Google Scholar - Sanjeev Kumar, Tara Chand,(2013).Formulation and Development of Floating and Mucoadhesive Microspheres of Clarithromycin ,The Pharma Innovation – Journal Vol. 2 No. 5
View at Publisher | View at Google Scholar - Mukesh P. Ratnaparkhi, Suresh B. Dhiwar, Kiran E. Dhage, Sharvari S. Bhore, Pratiksha M. Kadam and Pradeep S. Patil (2012) Formulation and in-vitro characterization of floating microspheres of Metfomin HCl ,Scholars Research Library Der Pharmacia Lettre,
View at Publisher | View at Google Scholar - Kapil Kumar and AK Ra, (2012). Development and Evaluation of Floating Microspheres of Curcumin, Tropical Journal of Pharmaceutical Research October 11 (5): 713-719
View at Publisher | View at Google Scholar - Parmar Kunal Vinodbhai a, Dr. M.C. Gohel b, Dr. R.K. Parikh b, Dr. Shital Bariyac Ms. Rajeshvari N. Suthar Sustained Release Floating Microspheres Of Acyclovir: Formulation, Optimization, Characterization And In Vitro Evaluation International Journal of D
View at Publisher | View at Google Scholar - Kumar Darapu B.N*, K. Sundaramoorthy And T.Vetrichelvan ,(2010) Formulation And In-Vitro Evaluation Of Gastroretensive Floating Microspheres Of Ranitidine Hydrochloride Vol.1 (2) Nov-Dec-An International Journal of Advances In Pharmaceutical Sciences
View at Publisher | View at Google Scholar - Singh Bandana, Kanoujia Jovita, Pandey Manisha*, Saraf Shubhini A.( 2010) Formulation and Evaluation of Floating Microspheres of Famotidine, International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.2, No.2, pp 1415-1420, April-
View at Publisher | View at Google Scholar - M.K. Deepaa, M. Karthikeyanb,(2009) Cefpodoxime Proxetil Floating Microspheres: Formulation and In Vitro Evaluation, Iranian Journal of Pharmaceutical Sciences Spring 5(2): 69-72.
View at Publisher | View at Google Scholar - Yuveraj Singh Tanwar, Pushpendra Singh Naruka, Garima Rani Ojha, (2007). Development and evaluation of floating microspheres of verapamil hydrochloride, Revista Brasileira de Ciências Farmacêuticas Brazilian Journal of Pharmaceutical Sciences vol. 43, n.
View at Publisher | View at Google Scholar - Rahaman Z, Ali M, Khar RK,(2006). Design and evaluation of bilayer floating tablets of captopril, Acta Pharma. 56(1): 49-57.
View at Publisher | View at Google Scholar - Tanwar YS, Naruka PS, Ojha GR,(2007).Development and evaluation of floating microspheres of verapamil hydrochloride, Brazilian J Pharm Sci. Vol:43(4): pg- 529-534.
View at Publisher | View at Google Scholar - Lee TW, Robinson JR, (2001).Remington: The Science and Practice of Pharmacy.20th Ed. Pennsylvania. Mack Publishing Company.
View at Publisher | View at Google Scholar - Naisarg D. Pujara, Namrata V. Patel Ankita P. Thacker, B rahmdutta K. Raval. Sumit M. Doshi, Ramesh B. Parmar, (2012). Floating Microspheres: A Noval Approach for Gastro Retention. World Journal Of Pharmacy and Pharmaceutical Sciences, Vol:1(3), pg-872-89
View at Publisher | View at Google Scholar - Shwetha Kamath K, Senthil Kumar S.K,(2012).Design and evaluation of floating microspheres of Rabeprazole Sodium, International Journal of Pharmacy and Pharmaceutical sciences, Vol: 4(3); Pg-357-367.
View at Publisher | View at Google Scholar - Anand Gadad, Chirag Naval, Krunal Patel, Panchaxari Dandagi and Vinaya.(2011). Formulation and evaluation of floating microspheres of captopril for prolonged gastric residence time, Indian Journal of Novel Drug Delivery, Vol:3(1), pg- 17-23.
View at Publisher | View at Google Scholar - Amol V. Pande, Pravin D. Vaidya, Aseem Arora, Madhura V. Dhoka,(2010) In-vitro and In-vivo evaluation of ethyl cellulose floating microspheres of cefpodoxime proxetil, International Journal of Pharmaceutical and Biomedical Research, Vol:1(4), pg-122-128.
View at Publisher | View at Google Scholar - Patil Hemangi S, Patil Moreshwar P, Tekade Bharat W, Thakare Vinod M,and Vijay R ,( 2009) Formulation and In-Vitro Evaluation of Floating Microspheres of Acyclovir, Arch Pharm Sci & Res, 1(1), 194 -198.
View at Publisher | View at Google Scholar - Deepaa M.K. and Karthikeyanb M, (2009).Cefpodoxime Proxetil Floating Microspheres: Formulation and In Vitro Evaluation, Iranian Journal of Pharmaceutical Sciences, 5(2), 69-72.
View at Publisher | View at Google Scholar - Srivastava Anand Kumar, Ridhurkar Devendra Narayanrao, Wadhwa Saurabh,(2005). Floating microspheres of cimetidine: Formulation, characterization and in vitro evaluation, Acta Pharm, 55, 277–285.
View at Publisher | View at Google Scholar - Sanjay S. Patel, S.Ray and R.S. Thakur, Formualtion and Evaluation of Floating Drug Delivery System containing Clarithromycin for Helicobacter Pylori, Acta Poloniae Pharmaceutica - Drug Research, 63(1), 53-61.
View at Publisher | View at Google Scholar - Patel Asha, Ray Subhabrata and Thakur Ram Sharnagat,(2006).In Vitro Evaluation and Optimization of Controlled Release Floating Drug Delivery System of Metformin Hydrochloride, DARU, 14( 2), 57-64.
View at Publisher | View at Google Scholar - Chien YW. (1992) Novel drug delivery system, Marcel Dekker Inc Publications: New York, 2(50): 16172.
View at Publisher | View at Google Scholar - Khar RK, Vyas SP, (2002).Targeted and controlled drug delivery novel carrier system, CBS Publishers and Distributors: New Delhi, 1: 417-54.
View at Publisher | View at Google Scholar - Arora S, Ali J, Ahuja A, Khar RK, Baboota S,( 2005). Floatng drug delivery systems: A review, AAPS Pharm Sci Tech 6(3): 372-390.
View at Publisher | View at Google Scholar - Wahlstrom B, Blennow G, (1978). A study on the fate of curcumin in the rat, Acta Pharmacol Toxicol, 43: 86-92.
View at Publisher | View at Google Scholar - Anand P, Kunnumakkara AB, Newman RA.( 2007). Bioavailability of Curcumin: Problems and Promises, Mol Pharmaceutics, 4 (6): 807-818.
View at Publisher | View at Google Scholar - Srivastava A, (2005). Floating microspheres of cimetidine: formulation, characterization and in vitro evaluation, Acta Pharm, 55: 277–285.
View at Publisher | View at Google Scholar - Maheswari U, Jain S, Bhadra D, Jain NK,(2003).Floating microspheres bearing acetohydroxamic acid for the treatment of H. pylori, J Pharm Pharmacol, 55: 1607-1613.
View at Publisher | View at Google Scholar - Higuchi WI. (1962).Analysis of data on the medicament release from ointments, J Pharm Sci, 51:802–804.
View at Publisher | View at Google Scholar - Korsmeyer RW, Gurny R, Doelker EM, Buri P, Peppas NA,(1983).Mechanism of solute release from porous hydrophilic polymers, Int J Pharm, 15: 25-35.
View at Publisher | View at Google Scholar - Rahman MH, Telny T, Kumaraswamy K. (2010).Comparative evaluation of HPMC K100 and poloxamer 188 - influence on release kinetics of Curcumin in floating microspheres, R J Pharm Chem Biol Sci, 1(2): 28-34.
View at Publisher | View at Google Scholar - G.K. Jani, M. C, Gohel, (1997).Journal of Controlled Release, 43,245 250.
View at Publisher | View at Google Scholar - Shashikant D. Barhate1, et al, Formulation and Evaluation of Floating Microspheres of Ketorolac Trometamol, IJPRD/2009/Pub/Arti/Vov-1/Issue-9/Nov/005.
View at Publisher | View at Google Scholar - Yogesh S Gattani, Durgacharan A Bhagwat, Akhil P Maske, (2008).Asian Journal of Pharmaceutics. October December.
View at Publisher | View at Google Scholar - Asha patel, subhabrata rayra, et al., DARU, 2006, 14(2).
View at Publisher | View at Google Scholar - M. Saravanan, K. Bhaskar, G. Srinivasa Rao and M.D. Dhanaraju. J. Microencapsulation 2003, 20 (3), 289 – 02.
View at Publisher | View at Google Scholar - Iannuccelli V, Coppi G, et al., Int J Pharm, 1998, 174; 47 – 54.
View at Publisher | View at Google Scholar - Lee J H, Park T G, Choi H K. J Microencapsules, 1999, 16(6), 715 – 729.
View at Publisher | View at Google Scholar - Streubel A, Siepamann I, Bodmeier R, Int J Pharm, 2002, 241, 279 – 82.
View at Publisher | View at Google Scholar - Ravi Kumar, Swati Patil, (2009).International Journal of Chem Tech Research, Oct-Dec Vol.1, No.4, pp 815- 825.
View at Publisher | View at Google Scholar - Arora S, Ali J, Ahuja A, Khar R.K., Baboota S, (2005). Floating Drug Delivery Systems: A Review. AAPS PharmSciTech, 06(03): E372-E390.
View at Publisher | View at Google Scholar - Talukder, R., Fassihi R, (2004). Gastroretentive drug delivery system: A mini review. Drug Dev. Ind. Pharm., Vol.30, pp.1019-1028.
View at Publisher | View at Google Scholar - Mr. Tanwar Y.S, (2006).Floating microspheres: Develpoment, Characterization and applications, Pharmainfo.net, Vol. 4, Issue 3.
View at Publisher | View at Google Scholar - Gaba Punam, Floating microspheres: A Review, Pharm info. net ,2008,Vol. 6 , issue 5.
View at Publisher | View at Google Scholar - Merck manual online drug library and www. merck.com
View at Publisher | View at Google Scholar - Uchida T, Yoshida K, Goto S, (1996). Preparation and characterization of poly-lactic acid microspheres containing water soluble dyes using a novel w/o/w emulsion solvent evaporation method. J Microencapsulation. 13;219-228.
View at Publisher | View at Google Scholar - Meral Yuce, Kandemir canefe, (2008).Indomethacinloaded microspheres: preparation, characterization and in-vitro evaluation regarding ethyl cellulose matrix material, Turk J. Pharm. Sci, 5 (3) 129-142.
View at Publisher | View at Google Scholar - Aphale s, shinde s, (2011) Development and evaluation of hollow microspheres of clarithromycin as a gastroretentive drug delivery system using eudragit polymers, Int. J. of Pharma and Bio Scie., 2, 344-358.
View at Publisher | View at Google Scholar - Tejaswi BS, Formulation and in vitro evaluation of clarithromycin floating microspheres for eradication of helicobacter pylori, Der Pharmacia Lettre, 3, 2011,90-101.
View at Publisher | View at Google Scholar - Pant P, Bansal K, and Rao RT, (2011). Formulation design and evaluation of gastroretentive mucoadhesive microspheres of clarithromycin, IJRPC 1, 347-351.
View at Publisher | View at Google Scholar - Sarojini AS, Kumar AP, and D Pradeep,(2010).Formulation and evaluation of albumin– chitosan floating microsphere containing clarithromycin and estimation by spectrophotometric method, RJPBCS, 1, 207-214.
View at Publisher | View at Google Scholar - Srivastava, A. K.; Ridhurkar, D. N., Wadhwa, S,( 2005). Floating microspheres of cimetidine: formulation, characterization and in vitro evaluation. Acta Pharm., v.55, p.277–285.
View at Publisher | View at Google Scholar - Thummel, K. E.; Shen, D. D, (2020).Design and optimization of dosage regimens: pharmacokinetic data. In: Hardman, J. G.; Limbirel, L. E.; Gilman, A. G., eds. Goodman and Gilman’s The pharmacological basis of therapeutics. New York: Mc. Graw Hill, 2001.
View at Publisher | View at Google Scholar - V. S. Mastiholimath, P. M. Dandagi, a. P. Gadad, Rashmi mathews, & a. R. Kulkarni,(2008) In vitro and in vivo evaluation of ranitidine hydrochloride ethyl cellulose floating micro particles, Journal of Microencapsulation. 25(5): 307–314.
View at Publisher | View at Google Scholar - Wakode RR, Bajaj AN (2008), Formulation and characterization of pramipexole loaded microspheres.
View at Publisher | View at Google Scholar - BC Behera, SK Sahooa, S Dhala, BB Barika, BK Guptab ,(2008).Characterization Of GlipizideLoaded Polymethacrylate Microspheres Prepared By An Emulsion Solvent Evaporation Method, Tropical Journal of Pharmaceutical Research, 2008; 7 (1): 879-885.
View at Publisher | View at Google Scholar - Yasunori Sato, Yoshiaki Kawashima, Hirofumi Takeuchi,(2003).Physicochemical propertie to determine the buoyancy of hollow microspheres (microballonss) prepared by the emulsion solvent diffusion method. European journal of pharmaceuticsand biopharmaceutics
View at Publisher | View at Google Scholar - Soppimath KS, Kulkarni AR, Rudzinski WE, Aminabhavi TM,(2002). Microspheres as floating drug-delivery systems to increase gastric retention of drugs, Drug Metab Rev 33:149-60.
View at Publisher | View at Google Scholar - Muthusamy K, Govindarazan G, Ravi TK, (2005). Preparation and evaluation of lansoprazole floating micropellets Indian Journal of Pharmaceutical Sciences 67:75-9.
View at Publisher | View at Google Scholar - Thanoo C, Sunny MC,Jayakrishnan A. (1993).Oral sustained-release drug delivery systems using polycarbonate microspheres capable of floating on the gastric fluid, J Pharm Pharmacol;45:21-4.
View at Publisher | View at Google Scholar - Joseph NJ, Lakshmi S, Jayakrishnan A, (2002). A floating-type oral dosage form for piroxicam based on hollow polycarbonate microspheres: In vitro and in vivo evaluation in rabbits. J Control Release; 79:71-9.
View at Publisher | View at Google Scholar - Stithit S, Chen W, Price JC, (1998). Development and characterization of buoyant theophylline microspheres with near zero order release kinetics, J Microencapsul 15:725-37.
View at Publisher | View at Google Scholar - Lee JH, Park TG, Choi HK, (1999). Development of oral drug delivery system using floating microspheres, J Microencapsul 16:715-29.
View at Publisher | View at Google Scholar - Gladiziwa U, Klotz U,(1993).Pharmacokinetics and pharmacodynamics of H2 receptor antagonists in patients with renal insufficiency, Clin Pharmacokinet 24:319-32.
View at Publisher | View at Google Scholar - Martin A and Swarbrick J,(1993).Physical pharmacy, 3rd edition, Mumbai: Varghese publishing house,444- 447.
View at Publisher | View at Google Scholar - Indian pharmacopoeia. Monographs on dosage forms. The Indian pharmacopoeia commission, Ghaziabad. [2007];3:182-183.
View at Publisher | View at Google Scholar - Indian pharmacopoeia. Monographs on dosage forms. The Indian pharmacopoeia commission, Ghaziabad. 2007;3:177-178.
View at Publisher | View at Google Scholar - Indian pharmacopoeia. Monographs on dosage forms. The Indian pharmacopoeia commission, Ghaziabad. 2007;3:183-184.
View at Publisher | View at Google Scholar - USP 27/NF 22, Asian edition, General test procedures, U.S. Pharmacopoeial convention, Rockville MD. 2000;1677- 1678.
View at Publisher | View at Google Scholar - Battue SK, Repay MA, Maunder S and Rio M Y.( 2007) Formulation and evaluation of rapidly disintegrating tablet Fenoverine tablets: Effect of superdisintegrants, Drug Dev Ind Pharm. 33:1225- 1232.
View at Publisher | View at Google Scholar - Sunada H, Yonezawa Y, Danjo K,Otsuka A and Iida K.( 1996). Preparation and evaluation of a compressed tablet rapidly disintegrating in the oral cavity, Chem Pharm Bull (Tokyo). 44: 2121-2127.
View at Publisher | View at Google Scholar - Basu S, Adhiyaman R (2008). Preparation and characterization of nitrendipine loaded eudragit RL100 microspheres prepared by an emulsion-solvent evaporation method, Trop J Pharmaceut Res 7: 1033-1041.
View at Publisher | View at Google Scholar - Yang Y, Chung T, Bai X, Chan W (2000). Effect of preparation conditions on morphology and release profiles of biodegradable polymeric microspheres containing protein fabricated by double emulsion method. Chem Eng Sci 55: 2223-2236.
View at Publisher | View at Google Scholar - Atyabi F, Mohammadi A, Dinarvand R, (2005).Preparation of nimodipine loaded microspheres: Evaluation of parameters, Iran J Pharm Sci 1: 143-152.
View at Publisher | View at Google Scholar - Basu S, Kavitha K, Rupeshkumar M (2010).Evaluation of ketorolac tromethamine microspheres by chitosan/gelatin complex coacervation, J Pharm Pharm Sci 78: 79-92.
View at Publisher | View at Google Scholar - Freiberg S, Zhu X (2004). Polymer microspheres for controlled drug release, Int J Pharm 282: 1-18.
View at Publisher | View at Google Scholar - Nighute A, Bhise S, (2009).Preparation and evaluation of rifabutin laded polymeric microspheres, Res J Pharm Tech 2: 371-374.
View at Publisher | View at Google Scholar - Guyot M, Fawaz F, (1998).Nifedipine loaded-polymeric microspheres: preparation and physical characteristics, Int J Pharm 197: 61-74.
View at Publisher | View at Google Scholar - Das M, Maurya D, (2008).Evaluation of diltiazem hydrochloride-loaded mucoadhesive microspheres prepared by emulsification internal gelation technique. Acta Pol Pharm-Drug Res 65: 249-259.
View at Publisher | View at Google Scholar - Nath B, Nath L, Mazumder B, Kumar P, (2010). Preparation and characterization of salbutamol sulphate loaded ethyl cellulose microspheres using water-in-oiloil emulsion technique, Iran J Pharm Res 9: 97-105.
View at Publisher | View at Google Scholar - Anand kumar, srivastava devendra, narayanrao ridhurkarsaurabh wadhwa, (2005). Floating microspheres of cimetidine; Formulation, characterization and in vitro evaluation: Acta Pharm 55: 277–285.
View at Publisher | View at Google Scholar - Arai, K. Kineemaki T, Fujita T, Bull Tokai, Reg. Fish.Res.Lab; 1968: 56-89.
View at Publisher | View at Google Scholar - Arjun Sony, Sonam Jain, (2013). Formulation & evaluation of floating microspheres of flupirtine Maleate; international journal of pharmacy & life sciences: Int. J. Of Pharm. & Life Sci 4(4): 2535-2540
View at Publisher | View at Google Scholar - Arul B, Kothai R., Sangameswaran B, Jayakar B, (2003). Formulation and Evaluation of Microspheres Containing Isoniazid; Indian Journal of Pharmaceutical Sciences: 65 (6): 640-642.
View at Publisher | View at Google Scholar - Hazner Dar S, (2004). Preparation and in vitro evaluation of Eudragit microspheres containing acetazolamide; International journal of pharmaceutics: 269: 131-140.
View at Publisher | View at Google Scholar
Dear Grace Pierce, Editorial Coordinator of Journal of Clinical Research and Reports, Thank you for the speedy and efficient peer review process. I appreciate the fact that your peer reviewers do not take months to respond like with some other journals. I would also like to thank the editorial office for responding quickly to my questions. It is an excellent journal. I plan to submit more manuscripts in the future. Best wishes from, Robert W. McGee

Robert W McGee
Dear Grace Pierce, Editorial Coordinator of Journal of Clinical Research and Reports, Working with you and your team on our recent publication in JCRR has been a truly wonderful and enjoyable experience. The responses were prompt, and the reviewers were patient, constructive, and highly professional. One reviewer in particular gave me the feeling that a professor was carefully reading and commenting on my coursework, which was deeply touching. The entire process was straightforward and hassle‑free, with no tedious online forms to complete. I highly recommend this journal. Best wishes from, DR Aibing Rao, Head of R&D

Aibing Rao
I Appreciate the Opportunity to Share my Experience with the Journal of Clinical Research and Reports. The peer review process was timely and constructive, and the feedback provided helped improve the quality of our manuscript. The editorial office was professional, responsive, and supportive throughout the process, ensuring smooth communication and efficient handling of the submission. Overall, it was a positive experience collaborating with your team.

Kashani Mehdi
Dear Mercy Grace, Editorial Coordinator of Obstetrics Gynecology and Reproductive Sciences, We would like to express our gratitude for your help at all stages of publishing and editing the article. The editors of the magazine answer all the necessary questions and help at every stage. We will definitely continue to cooperate and publish other works in the Obstetrics Gynecology and Reproductive Sciences! Best wishes from, Alla Konstantinovna Politova,

Alla Konstantinovna Politova
Dear Maria Emerson, Editorial Coordinator of International Journal of Clinical Case Reports and Reviews, What distinguishes International Journal of Clinical Case Report and Review is not only the scientific rigor of its publications, but the intellectual climate in which research is evaluated. The submission process is refreshingly free of unnecessary formal barriers and bureaucratic rituals that often complicate academic publishing without adding real value. The peer-review system is demanding yet constructive, guided by genuine scientific dialogue rather than hierarchical or authoritarian attitudes. Reviewers act as collaborators in improving the manuscript, not as gatekeepers imposing arbitrary standards. This journal offers a rare balance: high methodological standards combined with a respectful, transparent, and supportive editorial approach. In an era where publishing can feel more burdensome than research itself, this platform restores the original purpose of peer review — to refine ideas, not to obstruct them Prof. Perlat Kapisyzi, FCCP PULMONOLOGIST AND THORACIC IMAGING.
Perlat Kapisyzi
Dear Reader: We have published several articles in the Auctores Publishing, LLC, journal, Clinical Medical Reviews and Reports in recent years (CMRR). This is an ‘open access’ journal and the following are our observations. From the initial invitation to submit an article, to the final edits of galley proofs, we have found CMRR personnel to be professional, responsive, rapid and thorough. This entire process begins with Catherine Mitchell, Editorial Coordinator. She is simply outstanding, and, I believe, unparalleled in her capacity. I cannot imagine a more responsive and dedicated Editorial Coordinator. As I read the dates and timing of her correspondence with us, it seems that she never sleeps. I hope Auctores Publishing, LLC, appreciates her efforts as much as these authors do. Thank you to Auctores Publishing, LLC, to the Editorial Staff/Board, and to Catherine Mitchell from a grateful author(s).